A Case Report of Gestational Painless Thyroiditis
Keiichi Kamijo
Keiichi Kamijo*
Kamijo Thyroid Clinic and Kamijo Thyroid Research Institute, Sapporo, Japan
- *Corresponding Author:
- Keiichi Kamijo
Kamijo Thyroid Clinic and Kamijo Thyroid Research Institute, Sapporo, Japan
E-mail: katt@cocoa.ocn.ne.jp
Received date: May 07, 2018; Accepted date: May 11, 2018; Published date: May 23, 2018
Citation: Kamijo K (2018) A Case Report of Gestational Painless Thyroiditis. J Autoimmune Disord Vol 4:7. doi: 10.4172/2471-8513.100007
Copyright: © 2018 Kamijo K. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
A patient with previously diagnosed Graves’ hyperthyroidism had a transient episode of thyrotoxicosis late in gestation. This was associated with minimally palpable thyroid tissue, a rise in TgAb as well as TPOAb and negative TRAb(ECLIA) and TSAb(EIA). This case would suggest that patient with previously diagnosed Graves’ disease can have thyrotoxicosis without TRAb which is often found in painless thyroiditis occurring during the remission of Graves’ disease. In addition, an alternative cause for spontaneously resolving thyrotoxicosis was not discovered except painless thyroiditis. To our knowledge, this is the first case report of gestational thyrotoxic syndrome occurring late in gestation, but the specific mechanism has not been settled.
Keywords
Grave’s hyperthyroidism; Thyrotoxic; Thyroiditis; Thyrotoxicosis
Introduction
Painless or silent thyroiditis is a syndrome of thyrotoxicosis due to release of preformed thyroid hormones from disrupted thyroid follicle. The pathogenesis is frequently autoimmune and can further be subclassified into the sporadic and postpartum thyroiditis [1]. It was also reported that the thyrotoxic patients diagnosed as painless thyroiditis had previously had Graves’ hyperthyroidism, which was in remission [2-4]. To our knowledge, presented here is the first case report of painless thyroiditis occurring late in gestation who had a prior diagnosis of Graves’ disease.
Assays
Serum values of free triiodothyronine(FT3), free thyroxine(FT4), thyrotropin(TSH), TPOAb and TgAb levels were determined with electrochemiluminescence (Elecsys Free T3 assay, Elecsys Free T4 assay, Elecsys TSH assay, Elecsys anti-TPOAb and Elecsys TgAb assay (Roche Diagnostics GmbH, Penzberg, Germany) [5,6]. The reference limits (non-pregnant) were 2.00-4.40 pg/mL, 0.80-1.90 ng/dL, 0.45-4.50 μU/mL, and <52.0 IU/mL, <40.0 IU/mL, respectively. The TRAb (ECLIA) levels in the sera were measured with an inhibition assay kit-Elecsis ati-TSH receptor assay (Roche Diagnostics GmbH, Penzberg, Germany) [6] and TSAb(EIA), with TSAb kitYamasaEIA bioassay (Yamasa Corporation, Chiba, Japan), as previously described [5,6]. The reference limits were less than 2.0 IU/L and less than 120%. Vascularity index (VI) was measured using power Doppler sonography for differentiation between Graves’ disease and painless thyroiditis [6] and in the case of more than 80% of VI the diagnosis of Graves’ disease was made, as previously described.
Case Report
A 36-year old woman with a 5 year-history of treatment of Graves’ disease with thiamazole was first referred to our thyroid clinic in June 2012 for her follow-up. She was first diagnosed as Graves’ disease in 2007 by the initial doctor, although no data was available at that time. In April 2008, she was transferred to the next doctor for continuous maintenance therapy of Graves’ disease with thiamazole and she remained euthyroid and negative for TRAb. In June 2012, she was referred to our clinic with euthyroid state by the other day administration of thiamazole 5 mg daily. On physical examination, she had minimally palpable thyroid tissue without ophthalmopathy. In December 2012, thiamazole was discontinued and she went into a remission both by clinical and laboratory evaluation when she became pregnant. Her thyroid function test results during pregnancy and postpartum are summarized in Table 1. At 9 weeks of gestation, her thyroid function test showed TSH 2.67 μU/mL (normal 0.1-2.5), free T3 2.77 pg/mL, free T4 1.19 ng/dL and TRAb (ECLIA) <0.3 IU. She remained clinically and biochemically euthyroid between 11 and 20 weeks gestation. However, at 28 weeks of pregnancy, she suddenly complained of palpitation tiredness and hot intolerance. Her thyroid function test at this time confirmed the diagnosis of thyrotoxicosis based on suppressed TSH and elevated free T4 and T3. And she was strongly positive for TPOAb and TgAb. Because both TRAb (ECLIA) and TSAb (EIA) were negative. the recurrence of Graves’ disease was excluded. The primary diagnosis of unsuspected thyrotoxicosis which suddenly occurred in the third trimester was suspicious for painless thyroiditis, not Graves’ relapse. Furthermore, vascularity index measured using power Doppler sonography was normal at 64.2% (normal, 30-64.4) suggesting painless thyroiditis.
| Gestational time(weeks) and time(months) postpartum | Dates and events | FT3 (pg/mL) | FT4 (ng/dL) | TSH'μU/mL) | TRAb (ECLIA) IU/L) | TSAb (EIA) (%) | TPOAb (IU/mL) | TgAb (IU/mL) |
|---|---|---|---|---|---|---|---|---|
| 06/2012 MMI 5mg daily every other day | 3.04 | 1.31 | 1.2 | <0.3 | 146.1 | 1588.6 | ||
| 05/2013 MM was discontinued | 3.06 | 1.1 | 2.26 | <0.3 | ||||
| 9 weeks | Nov-13 | 2.77 | 1.19 | 2.67 | <0.3 | |||
| 11 weeks | Dec-13 | 2.87 | 1.05 | 1.69 | <0.3 | |||
| 20 weeks | Feb-14 | 2.85 | 1.21 | 0.87 | <0.3 | |||
| 28 weeks | 16/04/201 She developed palpitation, heat intolerance, malaise and increased bowel movement. | 9.21 | 3.38 | <0.01 | <0.3 | 92 | 137.6 | 832.5 |
| 30 weeks | 30/04/2014 | 6.18 | 2.55 | <0.01 | 0.4 | |||
| 32 weeks | May-14 | 2.89 | 1.35 | <0.01 | 0.4 | |||
| 36 weeks | Jun-14 | 1.86 | 0.8 | 0.13 | <0.3 | 97 | ||
| 06/2014 Delivery of the first child | ||||||||
| 2 months postpartum | Aug-14 | 3.07 | 1.19 | 1.04 | <0.3 | |||
| 6 months postpartum | Dec-14 | 2.65 | 1 | 1.64 | 0.3 | |||
| 12 months postpartum | Jun-15 | 3.03 | 1.14 | 2.24 | <0.3 | |||
Table 1: Laboratory Evaluation of Thyroid Function and Immunologic Markers over time in a case of Gestational Painless Thyroiditisa
As expected, thyrotoxicosis gradually and spontaneously resolved. Free T4 and T3 were dropped to the normal range, 2.89 pg/mL and 1.35 ng/dL at 32 weeks and at 1.86 pg/mL and 0.80 ng/dL, respectively. TSH level is undetectable at 32 weeks and slightly recovered to 0.13 μU/ml at 36 weeks and she became asymptomatic. Delivery was without complications and occurred at term. She remained euthyroid between 2 months and 16 months postpartum.
As a result, the diagnosis of painless thyroiditis in the late pregnancy was established and we have tentatively named it as ‘gestational painless thyroiditis’.
Discussion
To our knowledge, this is the first case report of gestational painless thyroiditis which occurred late in gestation. She had an unexpected thyrotoxicosis in late pregnancy and in view of the history of Graves’ disease, the recurrence of hyperthyroidism could not be excluded on clinical grounds alone. Usually the symptoms of Graves’ disease often improve during pregnancy and recur after delivery [7,8]. Of course, there is exceptions. Usually, a Tc-99m uptake or 24-hour radioactive iodine uptake is indicated as a means of distinguishing between painless thyroiditis and a relapse of hyperthyroidism. Of course, radioisotope scanning is absolutely contraindicated in pregnancy because the isotope may be transported through the placenta and concentrated by fetus. The TRAb test is of most value in their differential diagnosis. Since she was negative for both TRAb (ECLIA) and TSAb (EIA) at diagnosis, the recurrence of Graves’ disease can be excluded. Spontaneous resolution of thyrotoxicosis finally leads to the diagnosis of painless thyroiditis. In the present case thyrotoxicosis was spontaneously returning to a euthyroid state which was not followed by hypothyroid state. So, we compared the clinical course of painless thyroiditis in patients with Hashimoto’s thyroiditis and Graves’ disease which is in remission. Interestingly, out of 81 patients with painless thyroiditis occurring during remission of Graves’ disease, only 29 (36%) returned to euthyroid followed by hypothyroid phase.
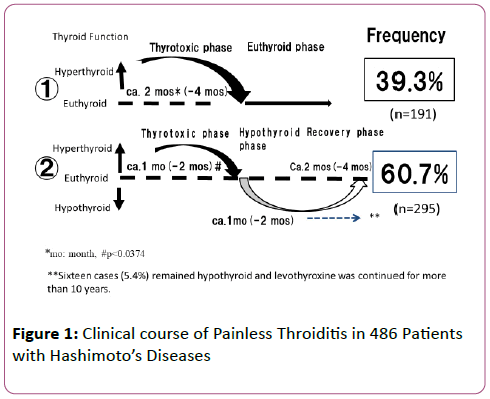
The remaining 52 cases never experienced hypothyroid phase in its clinical course like our case as shown in Figures 1 and 2. In contrast, about 60% of painless thyroiditis in patients with Hashimoto’s thyroiditis revealed an initial thyrotoxicosis followed by hypothyroid phase and the remaining 40% directly recovered to a euthyroid without hypothyroid phase [9]. In addition, the patients had not been taking any drugs including related to painless thyroiditis during pregnancy. Our case of transient thyrotoxicosis with painless thyroiditis occurring late in gestation in a patient with previous Graves’ hyperthyroidism has not been reported until now. Clinical aspects of human chorionic gonadotropin (hCG)-induced transient thyrotoxicosis [10] could not be thoroughly evaluated in this report, because hCG stimulated the maternal thyroid gland only in the early stages of gestation and often associated with hyperemesis gravidarum different from our case. The mechanism of gestational painless thyroiditis occurring late in pregnancy is not clear. We speculated that it may be due to the autoimmune process, because she had positive TPOAb and TgAb. Further studies will be needed to establish the exact mechanism.
In summary, including the current case report, painless thyroiditis can be subclassified into the 3 groups; sporadic, postpartum and gestational painless thyroiditis.
References
- Ginsberg J, Walfish PG (1997) Post-partum transient thyrotoxicosis with painless thyroiditis. Lancet 1: 1125-1128.
- Check JH, Avellino J (1980) Painless thyroiditis and transient thyrotoxicosis after Graves’ disease. Jama 244: 1361.
- Eckel RH, Green WL (1980) Postpartum thyrotoxicosis in a patient with Graves’ disease. Association with low radioactive iodine uptake, JAMA 243: 1454-1456.
- Kasayama S, Kitamura T, Inaba M, Otsuki M, Asanuma N, et al. (2018) Painless thyroiditis occurring during post-menopausal period, following remission of Graves’ disease. 31: 92-93.
- Kamijo K, Togashi K (2014) Development of more sensitive bioassay of TSAb due to the modification of conventional assay and its measurement in M22—seronegative Graves’ patients. Annals Thyroid Res 1: 5-10.
- Kamijo K (2010) Study on cutoff value setting for differentia; diagnosis between Graves’ disease and painless thyroiditis using the TRAb (Elecsys TRAb) measurement via the fully automated electrochemiluminescence immunoassay system. Endocr J 57: 895-902.
- Gardiner-Hill H (1929) Pregnancy complicating simple goiter and Graves’ disease. Lancet 1: 120-124.
- Astwood EB (1951) The use of antithyroid drugs during pregnancy. J Clin Endocrinol Metab 11: 1045-1056.
- Kamijo K (2006) Painless thyroiditis. Nihon Rinsho Review Suppl 1: 430-3.
- Swaminathan H, Chin RK, Lao TTH, Mak YT, Panesar NS, et al. (1989) Thyroid function in hyperemesis gravidarum. Acta Endocrinol 120: 155-160.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences